SESOIL Chemical Input Parameters
This month focuses on the SESOIL chemical input file. The chemical input file contains
information on physical properties of the contaminants. These properties are used by
the SESOIL pollutant cycle submodel to predict the transport and fate of contaminants in the
soil column. The large number of chemical input parameters means that SESOIL can be used for a
wide range of contaminant types. I have divided these parameters in two categories based on use.
Commonly used chemical parameters:
- Water solubility,
- Air and water diffusion coefficients,
- Henry′s Law Constant, and
- Organic carbon adsorption coefficient (Koc).
Rarely used parameters:
- Soil partition coefficient (Kd),
- Molecular weight,
- Valence,
- Biodegradation,
- Hydrolysis rate constants, and
- Ligand properties.
As SEVIEW 6.2 contains a 593 record database with 193 chemicals for the most part all you need to
do is find your chemical. Properties for additional contaminants can be found on the
internet or in various references. A detailed description of each input parameter is provided
below. Addition information on these parameters can be found in the SEVIEW help file and
User's Guide.
Commonly Used Parameters
These parameters are typically used when performing SESOIL modeling.
1.1 Water Solubility
Water solubility is the maximum aqueous concentration a substance can reach in mg/ml.
This property is temperature dependent and is typically measured at 25o C.
1.2 Diffusion Coefficients
Diffusion consists of the random motion of atoms or molecules in a gas or liquid. This motion
causes contaminants to disperse from areas of higher concentration to areas of lower concentration.
Diffusion is controlled by molecular weight, in that lighter atoms or molecules are dispersed
at higher rates. This dependency on weight can be used to establish diffusion coefficients for additional
chemicals using the equation below:
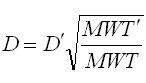 Where
Where
|
D =
|
Diffusion coefficient of the current molecule in cm2/sec
|
|
D′ =
|
Diffusion coefficient for a reference molecule in cm2/sec
|
|
MWT′ =
|
Molecular weight of the reference molecule in g/mole
|
|
MWT =
|
Molecular weight of the current molecule in g/mole
|
This equation can be used to estimate both air and water diffusion coefficients. Please note that
almost any substance can be used as the reference molecule. For instance the air and water diffusion
coefficients for 1,1,1-trichloroethane (TCA) are 0.078 cm2/sec and 8.80E-6 cm2/sec respectively.
The molecular weight for TCA is 133.4 g/mole.
1.2.1 Air Diffusion Coefficient
The air diffusion coefficient for a chemical in cm2/sec.
1.2.2 Water Diffusion Coefficient
The water diffusion coefficient for a chemical in cm2/sec. This parameter is not used by
SESOIL. It was added to SEVIEW 6.2 to simplify the link between SESOIL and AT123D. Addition
of this parameter means that users no longer need to edit the water diffusion coefficient
in AT123D prior to running the model.
1.3 Henry′s Law Constant
The Henry′s Law constant is the air:water partitioning coefficient in m3-atm/mol.
It relates the chemical concentrations between the gaseous and aqueous phases. The dimensionless
form is simply the ratio of the chemical concentration in air to that in water.
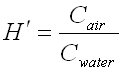 Where
Where
|
Cair =
|
Concentration in air in µg/L
|
|
Cwater =
|
Concentration in water in µg/L
|
|
H′ =
|
Dimensionless Henry′s Law constant
|
SESOIL uses the dimensional form of the Henry′s constant in m3-atm/mol. The dimensionless form
can be converted to the dimensional form using the equation below:
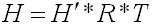 Where
Where
|
H =
|
Henry′s Law constant in m3-atm/mol
|
|
H′ =
|
Dimensionless Henry′s Law constant in
(µ/L)/(µ/L)
|
|
R =
|
Universal gas constant, in m3-atm/mol-K
= 8.2 E-5, and
|
|
T =
|
Temperature K = 298
|
1.4 Organic Carbon Adsorption Coefficient
The organic carbon adsorption coefficient (Koc) is the chemical adsorption coefficient normalized
to the organic carbon content of the soil in (µg/g)/(µg/ml). This parameter establishes the tendency
of a substance to bind to organic matter in the soil. SESOIL uses Koc and the soil organic carbon
content to calculate the soil partition coefficient (Kd).
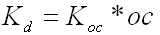 Where
Where
|
Kd =
|
Soil partition coefficient in (µg/g)/(µg/ml)
|
|
Koc =
|
Organic carbon adsorption coefficient in
(µg/g)/(µg/ml)
|
|
oc =
|
Fraction soil organic carbon content
|
Rarely Used Parameters
These parameters are not typically used when performing SESOIL modeling.
2.1 Soil Partition Coefficient
The distribution coefficient (Kd) of the compound on soil in (µg/g)/(µg/ml).
This is simply the Koc multiplied times the fraction of organic carbon content in the
soil. If a zero is entered for the soil partition coefficient, SESOIL will calculate a
Kd. Allowing SESOIL to perform this calculation ensures that the Kd value
is correct even if the soil organic carbon content value is changed in the soil input file.
2.2 Molecular Weight
Molecular weight is the sum of the atomic weights of the atoms. For example the molecular weight of benzene
(C6H6) which consists of six carbon and six hydrogen atoms is calculated below:
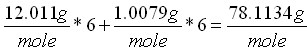 The atomic weights for carbon and hydrogen are 12.011 g/mole and 1.0079 g/mole respectively.
SESOIL only uses molecular weight if the complexation or cation exchange algorithms are used.
The atomic weights for carbon and hydrogen are 12.011 g/mole and 1.0079 g/mole respectively.
SESOIL only uses molecular weight if the complexation or cation exchange algorithms are used.
2.3 Biodegradation
Biodegradation is the process by which a chemical is converted to a new form via biotic
reactions. SESOIL simulates biodegradation as a first-order decay process. If used values
are generally based on site-specific measurements or
extremely low generic values.
2.3.1 Liquid Phase Biodegradation
Liquid phase biodegradation occurs to contaminants contained in soil moisture within
the vadose zone. In general the liquid phase biodegradation rate constant is higher
than in the solid phase.
2.3.2 Solid Phase Biodegradation
Solid phase biodegradation occurs to contaminants adsorbed on soil within the vadose zone.
2.4 Valence of the Chemical
The positive valence charge for a substance. Valence is controlled by the number
of electrons in the outermost orbital of an atom. Metals typically loose one or more valence electron
creating positive ions. These valence electrons control the type of chemical bonds and thus
formation of compounds. SESOIL uses valence along with the soil cation exchange capacity to
simulate the sorption of metals to soil.
2.5 Hydrolysis Rate Constants
Hydrolysis is a chemical reaction between salts of weak acids and bases with water. The
presence of these salts causes the water to ionize into negative hydroxyl ions (OH-)
and positive hydrogen ions (H+). This results in the formation of neutral, basic or
acidic solutions. Exactly which type of solution is formed depends on the properties of the salt ions.
2.5.1 Neutral
The neutral hydrolysis rate constant (L/mol/day).
2.5.2 Base
The base hydrolysis rate constant (L/mol/day).
2.5.3 Acid
The acid hydrolysis rate constant (L/mol/day).
2.6 Ligand Properties
Ligands are groups of molecules that attach to metal ions. Ligands (also known as
coordinating groups) basically take the place of an anion in a cation-anion molecule.
Such cation-ligand substances are called a complex.
2.6.1 Ligand Stability Constant
The ligand stability constant is the dimensionless ratio that relates the tendency of a
complex to dissociate. The more stable the complex the higher the ligand stability constant.
2.6.2 Moles Ligand per Mole Compound
The dimensionless ratio of the moles of ligand to the moles of the compound.
2.6.3 Molecular Weight of the Ligand
Like molecular weight the weight of the ligand is the sum of the atomic weights of the
atoms in g/mole. For example molecular weight of the ligand oxalato
(C2O42-) consists of two carbon and four oxygen atoms:
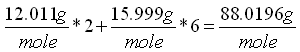 The atomic weights for carbon and oxygen are 12.011 g/mole and 15.9994 g/mole respectively.
The atomic weights for carbon and oxygen are 12.011 g/mole and 15.9994 g/mole respectively.
|
|
|